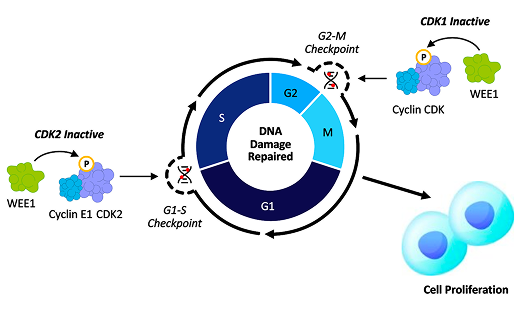
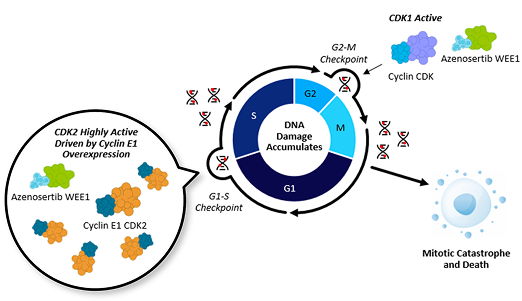
azenosertib mechanism of action.
In platinum resistance ovarian cancer, tumor cells are characterized by high proliferative rates and marked replication stress, commonly due to TP53 mutations and/or Cyclin E1 overexpression leading to a dysfunctional regulation at the G1/S checkpoint and premature entry into S phase. With this early checkpoint compromised, tumor cells become increasingly reliant on the G2/M checkpoint and on WEE1 to delay mitosis and preserve enough genomic integrity to survive.
When WEE1 is inhibited by azenosertib, the G2/M checkpoint is lost, forcing cells with unrepaired DNA damage to prematurely enter mitosis.2 This leads to mitotic catastrophe, accumulation of lethal DNA damage and ultimately tumor cell death.